SOLVED: Problem 1: Calculate the compressibility factor and the molar volume for ethane at 125°C and 24 bar using: i. The generalized Pitzer correlation using Lee and Kesler tables. ii. The Pitzer
5 (455) · $ 16.00 · In stock

VIDEO ANSWER: There is a chance that the pressure will be 300 bar and the temperature will be zero degree centigrade, so it will be a temperature of 3700 degrees. The T is equal to the number of moles. R is the universal gas constant, and the ideal
Numerade is a venture-backed, high-growth education technology startup based in Pasadena. We are singularly focused on creating exceptional video and interactive content experiences for education making the knowledge and skills of world class educators widely accessible and affordable to student audiences of all backgrounds. Our mission is to close the educational opportunity gap by unlocking and democratizing access to extraordinary educators and the content they have to offer.

SOLVED: In ethane (C2H6), Pc = 48.2 atm and Tc = 305.4 K. Calculate the pressure exerted by 74.8 g of C2H6 in a 200 cm3 container at 37.5°C using: A) The

Generalized Correlations for Gases (Lee-Kesler) - ppt video online download

Solved 7. (30 pts) Using the Lee-Kessler Correlation for

SOLVED: Problem 3 (35 marks): Calculate the compressibility factor Z and specific volume V cm/mole for ethane at 47.5°C and 25 bar by the following equations: 1. Ideal gas equation - 5

Solved Problem 3 (35 marks): Calculate the compressibility

Solved 1. Calculate the compressibity factor and the molar

Solved Compute a value of the coemcient 17. 0r ethane at

Solved Q1. Determine the molar volume of n-butane (Tc =425.1

Solved 2. (a) Calculate Z and V for ethylene at 25 °C and 12
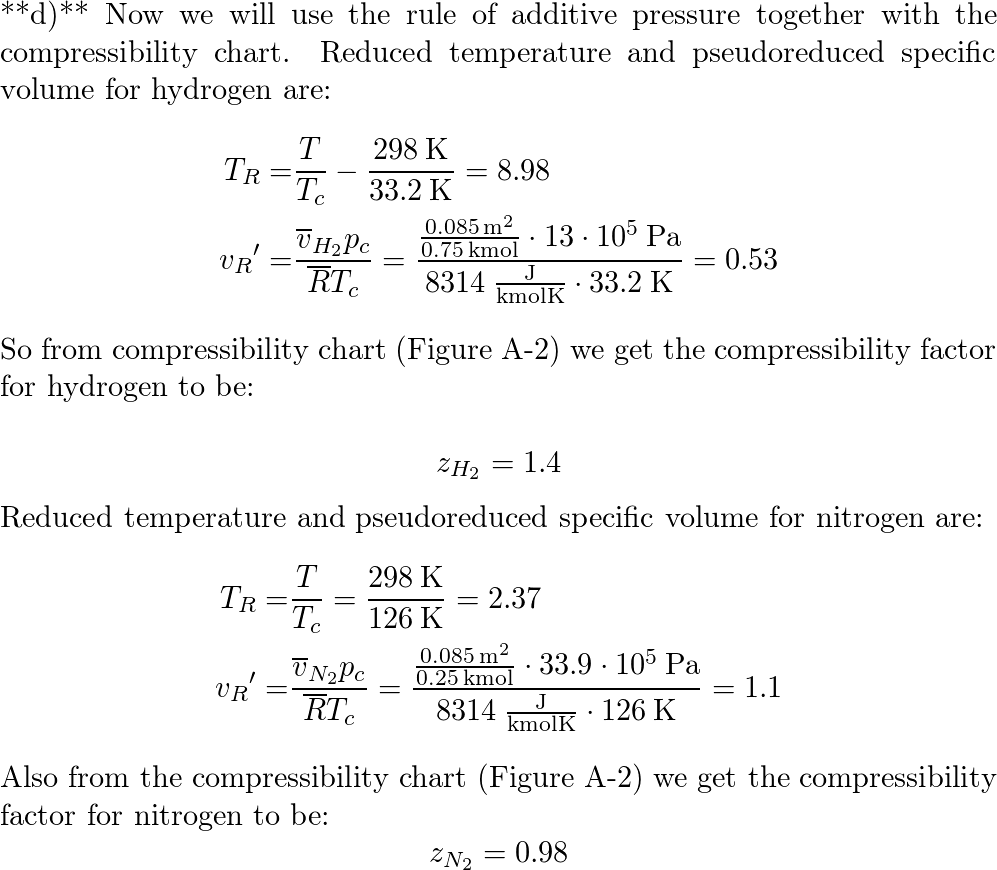
A gaseous mixture consisting of 0.75 kmol of hydrogen $(H_2)

A gaseous mixture consists of 75 percent methane and 25 perc

Calculate the molar volume and the compressibility

A tank contains 310 kg of a gaseous mixture of 70% ethane an

SOLVED: Problem 1: Calculate the compressibility factor and the molar volume for ethane at 125°C and 24 bar using: i. The generalized Pitzer correlation using Lee and Kesler tables. ii. The Pitzer
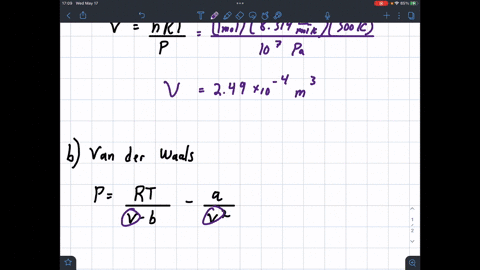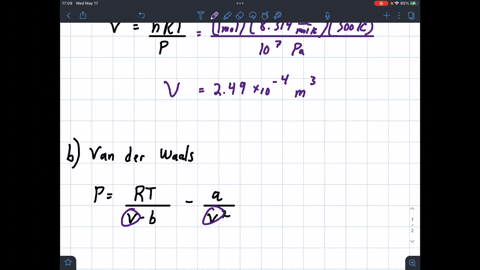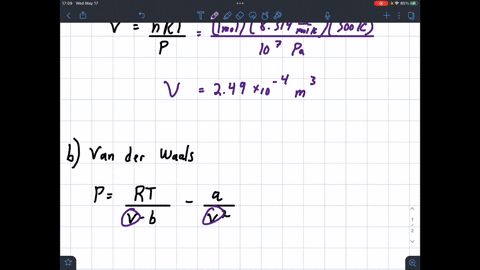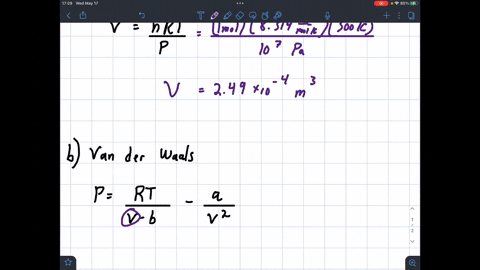
SOLVED: 1 Determine the molar volume of n-butane at 510 K and 25 bar based on each of the following: a) the ideal-gas state b) the generalized compressibility-factor correlation c) Pitzer correlations