total number of atoms in 44 g of Carbon dioxide is ?
4.5 (483) · $ 11.99 · In stock
Total number of atoms in 44 g of Carbon dioxide is

What mass of carbon is present in 44g of carbon dioxide? - Quora
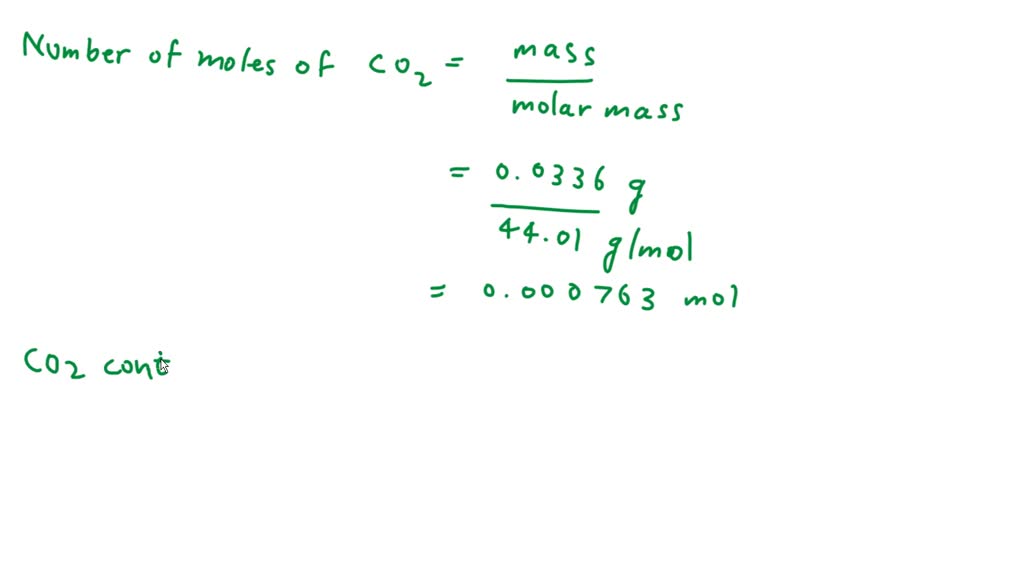
SOLVED: Atoms in 0.0336 g of CO2. The molar mass of CO2 is 44.01 g/mol. Find the number of moles of oxygen.
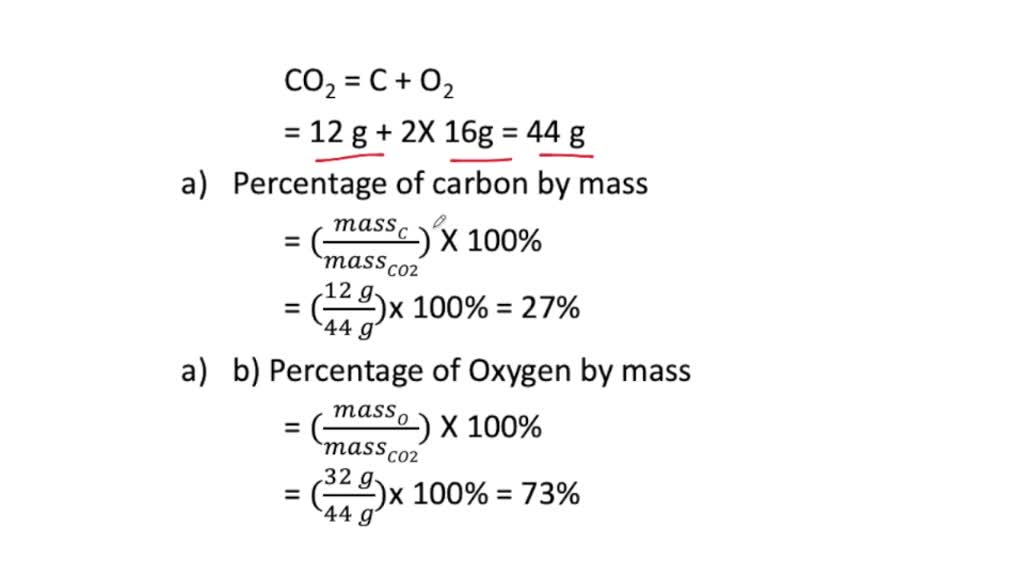
⏩SOLVED:a. What is the percent by mass of carbon in 44 g of carbon…
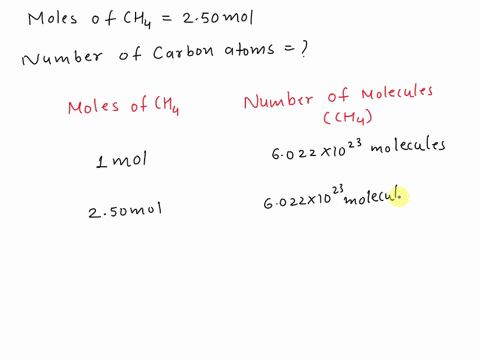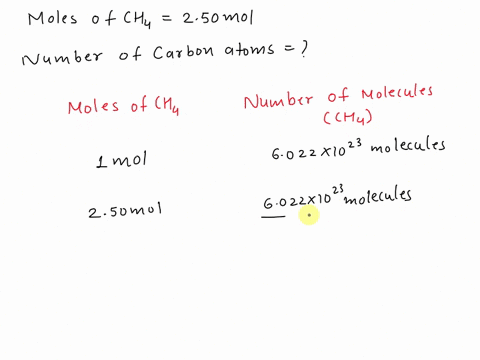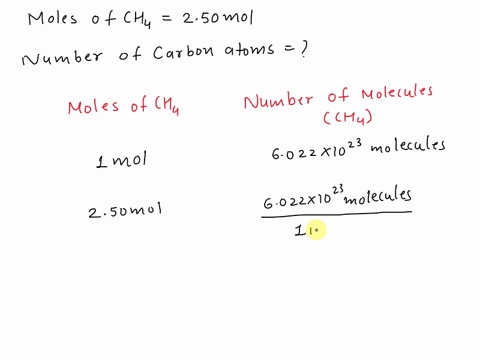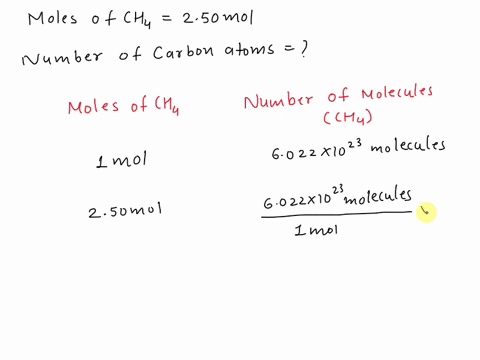
SOLVED: Assertion: Both 16g of CH4 and 44g of CO2 have the same number of atoms. Reason: Both contain 1g atom of carbon.
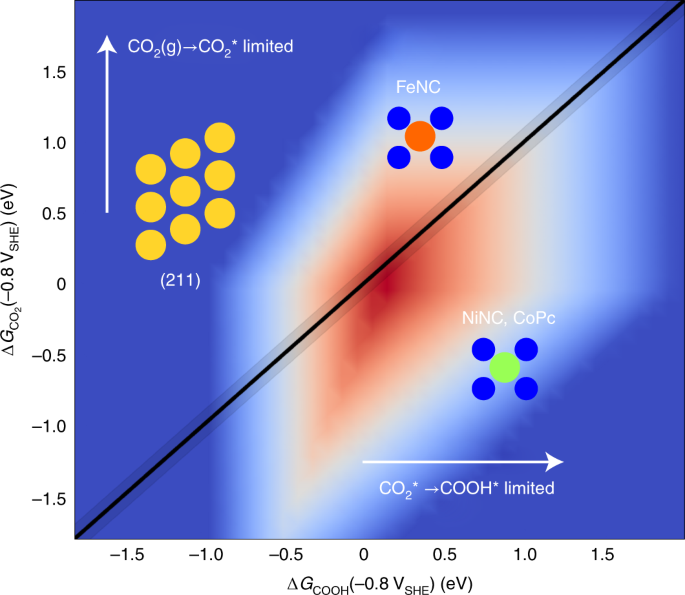
Unified mechanistic understanding of CO2 reduction to CO on transition metal and single atom catalysts

Solved Which of these contains the greatest number of atoms?

The total number of electrons in one molecular of carbon dioxide is
How many atoms of oxygen are contained in 44g of CO2? - Quora
total number of atoms in 44 g of Carbon dioxide is ?

The number of oxygen atoms in 4.4 g of CO(2) is approximately

Room-temperature photosynthesis of propane from CO2 with Cu single atoms on vacancy-rich TiO2

Total no. of atoms in 44 gm of CO_2 is –, CLASS 11, MOLE CONCEPTS, CHEMISTRY

What mass of carbon is present in 44g of carbon dioxide? - Quora

How many atoms of oxygen are contained in 44g of CO2? - Quora

SOLVED: How many grams of carbon dioxide, CO2, (MW = 44.01 g/mol) occupy a volume of 15.3 L at STP? Group of answer choices 0.638 g 0.348 g 0.683 g 30.1 g 7.79 g







