The compression factor (compressibility factor) for one mole of a Van der..
4.7 (474) · $ 16.99 · In stock

Solution For The compression factor (compressibility factor) for one mole of a Van der Waals' gas at 0∘C and 100 atmosphere pressure is found to be 0.5 . Assuming that the volume of a gas molecule is
The compression factor (compressibility factor) for one mole of a Van der Waals' gas at 0∘C and 100 atmosphere pressure is found to be 0.5 . Assuming that the volume of a gas molecule is negligible, calculate the Van der Waals' constant 'a',
[JEE 2001]
ns. 1.256 atm L2 mol−2
Z=1−VmRTaZ=RTPVm⇒Vm=PRTZZ=1−(RT)2a×ZP0.5=1−(273×0.0821)2×.5a×100a=1.256 atm L2 mol−2
Video solution 1: The compression factor (compressibility factor) for one mole of a Van der Waals' gas at 0∘C and 100 atmosphere pressure is found to be 0.5 . Assuming that the volume of a gas molecule is negligible, calculate the Van der Waals' constant 'a',
[JEE 2001]
ns. 1.256 atm L2 mol−2
Z=1−VmRTaZ=RTPVm⇒Vm=PRTZZ=1−(RT)2a×ZP0.5=1−(273×0.0821)2×.5a×100a=1.256 atm L2 mol−2

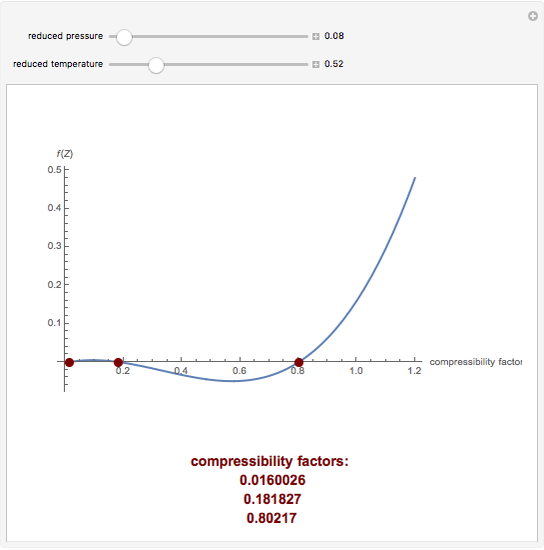
al Gases f.a What is the compressibility factor (Z) 0.02 mole of a
![Bengali] For one mole of a van der Waals' gas when b = 0and T =300 K](https://d10lpgp6xz60nq.cloudfront.net/physics_images/PAT_CHE_0XI_P03_C06_E20_001_Q01.png)
Bengali] For one mole of a van der Waals' gas when b = 0and T =300 K

Solved The compressibility factor can be expressed in terms

Welcome to Chem Zipper.com: The compressibility factor for 1
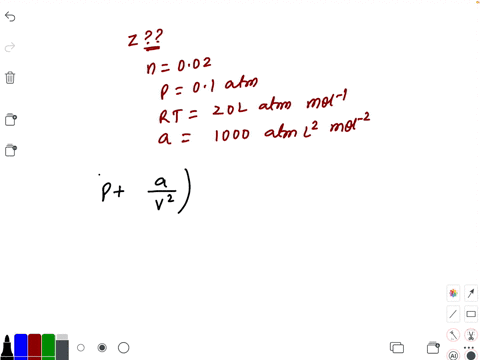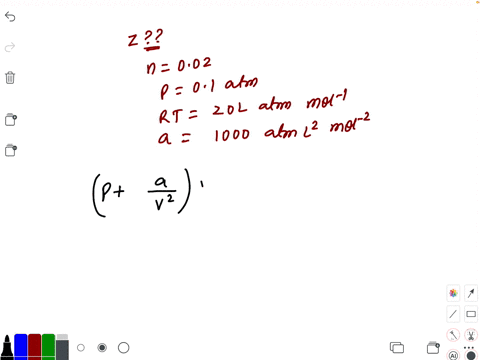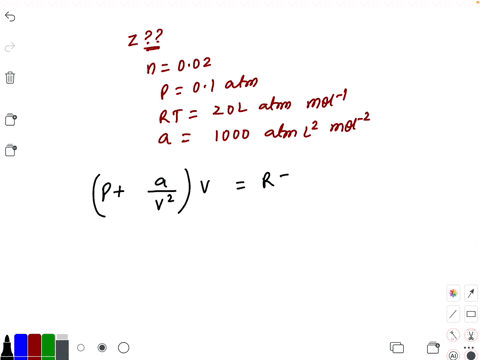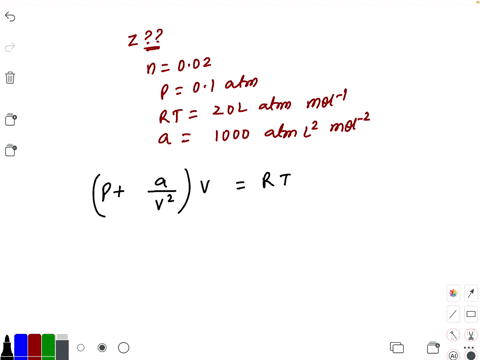
⏩SOLVED:Compressibility factor for 1 mol of a van der Waals gas

CBSE 11th Chemistry - State of Matter Three Marks Questions
![Bengali] The compresibility factor (Z) of one mole of a van der waals](https://static.doubtnut.com/ss/web/2552448.webp)
Bengali] The compresibility factor (Z) of one mole of a van der waals
![Bengali] What will the value of compressibility factor (Z) be for a g](https://static.doubtnut.com/ss/web/2601444.webp)
Bengali] What will the value of compressibility factor (Z) be for a g
![Bengali] The compressibility factor (Z) of one mole of a van der Waal](https://static.doubtnut.com/ss/web-overlay-thumb/9067415.webp)
Bengali] The compressibility factor (Z) of one mole of a van der Waal

Sheet - 01 - Real Gas, PDF, Gases

The compression factor (compressibility factor) for 1 mol of a van der