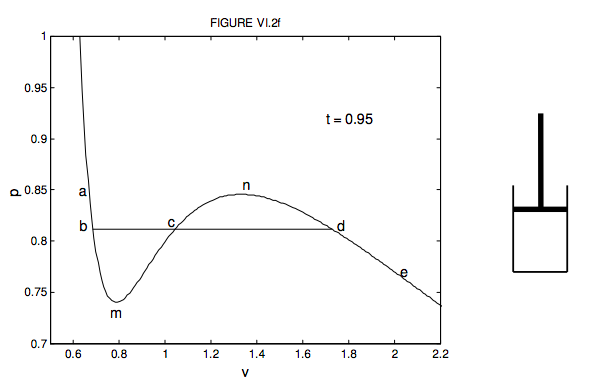
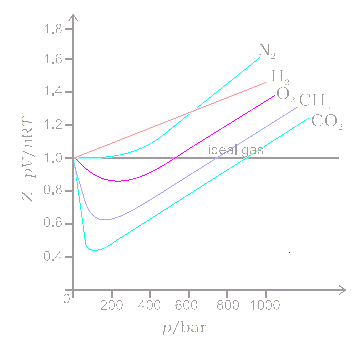
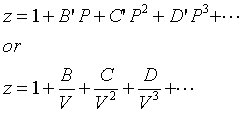Compressibility factor (Z) for a van der Waals real gas at
4.5 (402) · $ 26.50 · In stock

Share your videos with friends, family and the world

What is the major factor that accounts for most of the difference in these two values of pressure (ideal gas law vs. van der Waals equation)?

Class Notes on Compressibility of a Real Gas, CH 417, Study notes Physical Chemistry
COMPREHENSION_TYPE from IIT-JEE PREVIOUS YEAR (CHEMISTRY) STATES OF MATTER for Class 12
Complete Solutions to Mock Test 1 of chapter MOCK TEST of Class 11 book with complete answers and questions

The compressibility factor a real gas high pressure is:-1 - frac{Pb} {RT}1 + frac {RT} {Pb}11 + frac {Pb} {RT}
6.3: Van der Waals and Other Gases - Physics LibreTexts

THE 3rd STATE OF MATTER – What is a Real Gas? – Computer Aided Design & The 118 Elements

Deviation From Ideal Gas Behavior - Study Material for IIT JEE

Compressibility of a van der Waals Gas, Physical Chemistry I

If `Z` is a compressibility factor, van der Waals' equation at low pressure can be written as

Non-ideal behavior of gases (article)
![Bengali] In terms of critical constants, the compressibility factor i](https://static.doubtnut.com/ss/web/9704875.webp)
Bengali] In terms of critical constants, the compressibility factor i
Van der waals equation: Derivation, Explanation
If Z is a compressibility factor, van der Waals' equation at low pressure can be written as - Sarthaks eConnect

Real Gases and the Virial Equation